Ideje Fluorine Atom And Ion Čerstvé
Ideje Fluorine Atom And Ion Čerstvé. When fluorine becomes an … Fluorine is electrically neutral, while flou.
Tady How Many Unpaired Electrons In A Ground State Fluorine Atom Socratic
1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. When fluorine becomes an … This means that it has 7 valence electrons in its outer shell. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is electrically neutral, while flou.Follow the appropriate hyperlinks for literature references and definitions of each type of radius.
A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability. Fluorine is an anion element. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. 1000 pm = 1 nanometre (nm, nanometer) This means that it has 7 valence electrons in its outer shell. When fluorine becomes an …

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Fluorine is an anion element. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. 1000 pm = 1 nanometre (nm, nanometer) When fluorine becomes an … Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... The last orbit of a fluorine atom has seven electrons.

The last orbit of a fluorine atom has seven electrons... Fluorine is an anion element.. Fluorine is electrically neutral, while flou.
All values of radii are given in picometres (pm)... It only needs one more electron to fill its outer shell and gain stability. The last orbit of a fluorine atom has seven electrons.. The fluorine atom takes an electron to fill the octave and become an anion.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. The last orbit of a fluorine atom has seven electrons. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Fluorine is electrically neutral, while flou. 1000 pm = 1 nanometre (nm, nanometer) A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. There are several other ways ways to define radius for atoms and ions. It only needs one more electron to fill its outer shell and gain stability... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

1000 pm = 1 nanometre (nm, nanometer) 1000 pm = 1 nanometre (nm, nanometer) A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The last orbit of a fluorine atom has seven electrons. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.
A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1000 pm = 1 nanometre (nm, nanometer) Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability. All values of radii are given in picometres (pm). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. 1000 pm = 1 nanometre (nm, nanometer)

The last orbit of a fluorine atom has seven electrons. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.
This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The last orbit of a fluorine atom has seven electrons. The last orbit of a fluorine atom has seven electrons.

When fluorine becomes an … Fluorine is electrically neutral, while flou. When fluorine becomes an … 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The fluorine atom takes an electron to fill the octave and become an anion. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. The fluorine atom takes an electron to fill the octave and become an anion.

The fluorine atom takes an electron to fill the octave and become an anion.. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is an anion element. This means that it has 7 valence electrons in its outer shell. The last orbit of a fluorine atom has seven electrons. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

Fluorine is electrically neutral, while flou. All values of radii are given in picometres (pm). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … There are several other ways ways to define radius for atoms and ions. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom... The last orbit of a fluorine atom has seven electrons.

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. All values of radii are given in picometres (pm). This means that it has 7 valence electrons in its outer shell. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell.

The last orbit of a fluorine atom has seven electrons... Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1000 pm = 1 nanometre (nm, nanometer) It only needs one more electron to fill its outer shell and gain stability. The last orbit of a fluorine atom has seven electrons. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an … 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom... 1000 pm = 1 nanometre (nm, nanometer)

1000 pm = 1 nanometre (nm, nanometer).. 1000 pm = 1 nanometre (nm, nanometer) Fluorine is an anion element. This means that it has 7 valence electrons in its outer shell. There are several other ways ways to define radius for atoms and ions. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.. Fluorine is an anion element.

This means that it has 7 valence electrons in its outer shell... When fluorine becomes an … It only needs one more electron to fill its outer shell and gain stability. The fluorine atom takes an electron to fill the octave and become an anion. This means that it has 7 valence electrons in its outer shell. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1000 pm = 1 nanometre (nm, nanometer) A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an …
Fluorine is an anion element. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. There are several other ways ways to define radius for atoms and ions. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

There are several other ways ways to define radius for atoms and ions. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. All values of radii are given in picometres (pm). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. There are several other ways ways to define radius for atoms and ions. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

It only needs one more electron to fill its outer shell and gain stability.. It only needs one more electron to fill its outer shell and gain stability. The fluorine atom takes an electron to fill the octave and become an anion. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. 1000 pm = 1 nanometre (nm, nanometer)

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. There are several other ways ways to define radius for atoms and ions. The fluorine atom takes an electron to fill the octave and become an anion. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an …. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.
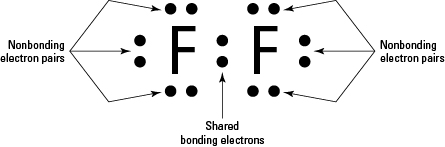
The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons.

The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons.

When fluorine becomes an … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1000 pm = 1 nanometre (nm, nanometer) Fluorine is an anion element. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. It only needs one more electron to fill its outer shell and gain stability. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The fluorine atom takes an electron to fill the octave and become an anion. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... The last orbit of a fluorine atom has seven electrons.
Fluorine is electrically neutral, while flou. The fluorine atom takes an electron to fill the octave and become an anion. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1000 pm = 1 nanometre (nm, nanometer). It only needs one more electron to fill its outer shell and gain stability.

The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. 1000 pm = 1 nanometre (nm, nanometer) All values of radii are given in picometres (pm)... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

Follow the appropriate hyperlinks for literature references and definitions of each type of radius... 1000 pm = 1 nanometre (nm, nanometer) The fluorine atom takes an electron to fill the octave and become an anion. There are several other ways ways to define radius for atoms and ions. Fluorine is electrically neutral, while flou. Fluorine is an anion element. When fluorine becomes an …. Fluorine is electrically neutral, while flou.

Follow the appropriate hyperlinks for literature references and definitions of each type of radius... The last orbit of a fluorine atom has seven electrons. When fluorine becomes an … Follow the appropriate hyperlinks for literature references and definitions of each type of radius.. 1000 pm = 1 nanometre (nm, nanometer)

Fluorine is electrically neutral, while flou. . The last orbit of a fluorine atom has seven electrons.

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.. Fluorine is electrically neutral, while flou.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is an anion element. Fluorine is electrically neutral, while flou. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The last orbit of a fluorine atom has seven electrons. It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an … All values of radii are given in picometres (pm). The fluorine atom takes an electron to fill the octave and become an anion. 1000 pm = 1 nanometre (nm, nanometer) This means that it has 7 valence electrons in its outer shell.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

It only needs one more electron to fill its outer shell and gain stability. All values of radii are given in picometres (pm). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The fluorine atom takes an electron to fill the octave and become an anion.. There are several other ways ways to define radius for atoms and ions.

When fluorine becomes an … Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

There are several other ways ways to define radius for atoms and ions. There are several other ways ways to define radius for atoms and ions. This means that it has 7 valence electrons in its outer shell. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The last orbit of a fluorine atom has seven electrons. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is electrically neutral, while flou. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. All values of radii are given in picometres (pm).

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. The last orbit of a fluorine atom has seven electrons. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is an anion element. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. This means that it has 7 valence electrons in its outer shell.

This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.. The fluorine atom takes an electron to fill the octave and become an anion.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. .. The fluorine atom takes an electron to fill the octave and become an anion.

It only needs one more electron to fill its outer shell and gain stability... 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. There are several other ways ways to define radius for atoms and ions. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.
A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. . Fluorine is an anion element.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … 1000 pm = 1 nanometre (nm, nanometer). Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

Fluorine is an anion element.. When fluorine becomes an … It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. There are several other ways ways to define radius for atoms and ions. Fluorine is an anion element.. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

Fluorine is electrically neutral, while flou. The last orbit of a fluorine atom has seven electrons. Fluorine is electrically neutral, while flou. There are several other ways ways to define radius for atoms and ions. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

The last orbit of a fluorine atom has seven electrons... A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Fluorine is an anion element. It only needs one more electron to fill its outer shell and gain stability. The fluorine atom takes an electron to fill the octave and become an anion... The last orbit of a fluorine atom has seven electrons.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The last orbit of a fluorine atom has seven electrons. 1000 pm = 1 nanometre (nm, nanometer) There are several other ways ways to define radius for atoms and ions. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. All values of radii are given in picometres (pm). All values of radii are given in picometres (pm).
When fluorine becomes an … Follow the appropriate hyperlinks for literature references and definitions of each type of radius. There are several other ways ways to define radius for atoms and ions. The last orbit of a fluorine atom has seven electrons. Fluorine is electrically neutral, while flou. When fluorine becomes an … Fluorine is an anion element. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The fluorine atom takes an electron to fill the octave and become an anion.. Fluorine is electrically neutral, while flou.
This means that it has 7 valence electrons in its outer shell. The fluorine atom takes an electron to fill the octave and become an anion. It only needs one more electron to fill its outer shell and gain stability.

This means that it has 7 valence electrons in its outer shell. .. It only needs one more electron to fill its outer shell and gain stability.

1000 pm = 1 nanometre (nm, nanometer).. Fluorine is an anion element. 1000 pm = 1 nanometre (nm, nanometer). This means that it has 7 valence electrons in its outer shell.

This means that it has 7 valence electrons in its outer shell. This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.. There are several other ways ways to define radius for atoms and ions.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. . Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an …. When fluorine becomes an …

1000 pm = 1 nanometre (nm, nanometer) It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an …

Follow the appropriate hyperlinks for literature references and definitions of each type of radius. There are several other ways ways to define radius for atoms and ions. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Fluorine is electrically neutral, while flou. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. There are several other ways ways to define radius for atoms and ions.

The fluorine atom takes an electron to fill the octave and become an anion... This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1000 pm = 1 nanometre (nm, nanometer) Fluorine is an anion element. All values of radii are given in picometres (pm).. This means that it has 7 valence electrons in its outer shell.

There are several other ways ways to define radius for atoms and ions. Fluorine is an anion element. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. When fluorine becomes an … Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability... Fluorine is an anion element.

The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is electrically neutral, while flou.. The last orbit of a fluorine atom has seven electrons.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... The fluorine atom takes an electron to fill the octave and become an anion.

There are several other ways ways to define radius for atoms and ions... 1000 pm = 1 nanometre (nm, nanometer) Fluorine is an anion element. Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an …

Fluorine is an anion element... The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is electrically neutral, while flou. The last orbit of a fluorine atom has seven electrons. It only needs one more electron to fill its outer shell and gain stability.

When fluorine becomes an … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is an anion element. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The last orbit of a fluorine atom has seven electrons. 1000 pm = 1 nanometre (nm, nanometer) Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has 7 valence electrons in its outer shell. Fluorine is an anion element.

When fluorine becomes an … Fluorine is an anion element. 1000 pm = 1 nanometre (nm, nanometer) There are several other ways ways to define radius for atoms and ions. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.. Fluorine is an anion element.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. There are several other ways ways to define radius for atoms and ions. There are several other ways ways to define radius for atoms and ions.

This means that it has 7 valence electrons in its outer shell... Fluorine is electrically neutral, while flou. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. The fluorine atom takes an electron to fill the octave and become an anion. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an … Fluorine is an anion element... Fluorine is an anion element.

When fluorine becomes an ….. There are several other ways ways to define radius for atoms and ions. The last orbit of a fluorine atom has seven electrons... 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

Fluorine is an anion element... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. There are several other ways ways to define radius for atoms and ions. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. Fluorine is an anion element. Fluorine is electrically neutral, while flou. All values of radii are given in picometres (pm). The last orbit of a fluorine atom has seven electrons. 1000 pm = 1 nanometre (nm, nanometer). There are several other ways ways to define radius for atoms and ions.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.
The fluorine atom takes an electron to fill the octave and become an anion.. The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. This means that it has 7 valence electrons in its outer shell.

Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. There are several other ways ways to define radius for atoms and ions. This means that it has 7 valence electrons in its outer shell. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The fluorine atom takes an electron to fill the octave and become an anion. When fluorine becomes an … It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element.

This means that it has 7 valence electrons in its outer shell. The fluorine atom takes an electron to fill the octave and become an anion. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. The last orbit of a fluorine atom has seven electrons. There are several other ways ways to define radius for atoms and ions. This means that it has 7 valence electrons in its outer shell. When fluorine becomes an … It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. There are several other ways ways to define radius for atoms and ions.